Nuclear Chemistry

This unit is about isotopes and how they decay. We will discuss how isotopes are different versions of an atom, their stability, and how they decay (alpha, beta, and gamma). We will also discuss the difference between nuclear fusion and nuclear fission and what roles these two process have on bombs, sources of energy and potential dangers.
The goal for this unit:
Students understand an isotope is an atom usually with extra neutrons in the nucleus.
The extra neutrons can make the atom unstable.
If the atom is unstable its nucleus will break apart releasing particles and energy (radioactive decay).
The particles can be helium (alpha), electrons (beta) or photons (gamma).
Nuclear Fussion: Atoms join together and release energy.
Nuclear Fission: Atom breaks apart to release energy (radioactive decay)
State Standard Covered:
H.1P.1 (B): Explain how the composition of the nucleus is related to isotopes and radioactivity.
The goal for this unit:
Students understand an isotope is an atom usually with extra neutrons in the nucleus.
The extra neutrons can make the atom unstable.
If the atom is unstable its nucleus will break apart releasing particles and energy (radioactive decay).
The particles can be helium (alpha), electrons (beta) or photons (gamma).
Nuclear Fussion: Atoms join together and release energy.
Nuclear Fission: Atom breaks apart to release energy (radioactive decay)
State Standard Covered:
H.1P.1 (B): Explain how the composition of the nucleus is related to isotopes and radioactivity.
Assignments, Labs, Homework and Study Guide to Download:
Available in word doc and in PDF
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Interesting Links
Cloud Chamber: Shows Helium atoms decaying off of Lead-210
Nuclear Chemistry: Explains what happens to an atom to make it radioactive.
Where does the sun get its energy? - 8 min video
Nuclear Reactor Explained -4 min video
Radiation effects on humans -5 min video
Bill Nye-Nuclear Radiation- 23 min
Nuclear bomb test: structures being destroyed, trees, etc.
Nuclear bomb tests: mult footage, put together with music, some good footage
Underground Nuclear Test:
H-Bomb: Ship for scale
Fusion & Fission: Compared in a nutshell, a 7 min video
Mouse traps: A simulation of nuclear fission, as one reactions causes others ...creating a chain reaction.
Sun Tsunami: Close up of the sun, and how fusion on the surface can kill your cell phone
Hiroshima: Bomb dropped on Japan during WWII
Trinity Atom Bomb: Shows explosion of test
24 Hours after Hiroshima: Part 1/3 - 15 min
24 Hours after Hiroshima: Part 2/3 - 15 min
24 Hours after Hiroshima: Part 3/3 - 15 min
Atomic Cafe: A documentary of the history of the atomic bomb 85 min video.
What happens in a nuclear Meltdown: Its in German but a great picture representation.
Nuclear Boy Scout: Boy scout builds a bomb
Web Quest Link - record the answers in your notebook
Alpha Decay
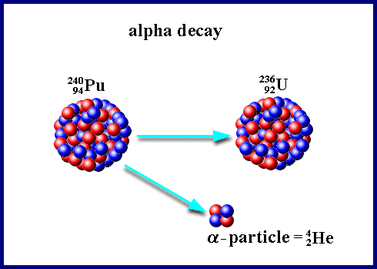
In Alpha Decay a helium atom is emitted from the nucleus of an atom. Notice that it's atomic mass goes down by 4 (lost 4 particles) and its atomic number goes down by 2 (lost 2 protons).
Beta Decay
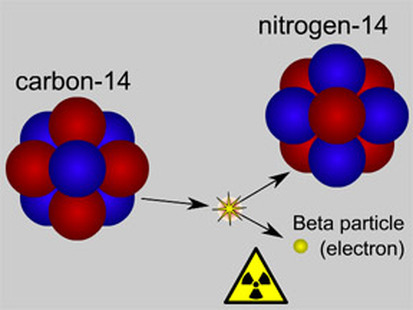
In Beta Decay, an ELECTRON is emitted as an atom decays. Inside your body, the Carbon 14 is decaying into Nitrogen 14. How this happens is.......a neutron in the nucleus of the carbon splits into a proton and electron ( neutrons are made of a proton and electron....yes weird...but a positive (+) and a negative (-) equals a neutral neutron.
So the carbon 14, with its 6 protons, 6 electrons, and 8 neutrons.....now turns into a nitrogen with 7 protons, 6 electrons, and 7 neutrons....the electron gets shot out and we detect is as radiation.
So the carbon 14, with its 6 protons, 6 electrons, and 8 neutrons.....now turns into a nitrogen with 7 protons, 6 electrons, and 7 neutrons....the electron gets shot out and we detect is as radiation.
Gamma Decay
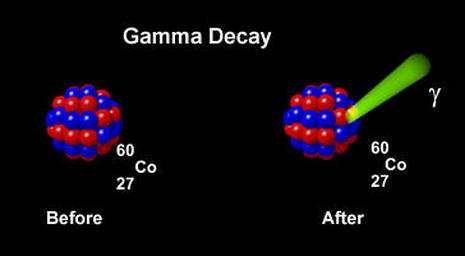
In Gamma Decay a gamma ray is emitted by the nucleus of an atom. The photon (packet of energy) of the gamma ray is at high energy levels. Notice NO particles are lost in gamma decay, only energy leaves the nucleus and thus the atom is at a lower energy state.