Chemical Reactions and Chemical Bonds

This unit is about chemical reactions, what happens during them, and what the signs of a chemical reactions. We will also looks at the different types of bonds, such as, Ionic, Polar Covalent, Nonpolar Covalent, and Metallic.
State Standards Covered:
H.1P.2 Describe how different types and strengths of bonds affect the physical and chemical properties of compounds.
H.2P.1 (A) Explain how chemical reactions result from the making and breaking of bonds in a process that absorbs or releases energy. (B) Explain how different factors can affect the rate of a chemical reaction.
State Standards Covered:
H.1P.2 Describe how different types and strengths of bonds affect the physical and chemical properties of compounds.
H.2P.1 (A) Explain how chemical reactions result from the making and breaking of bonds in a process that absorbs or releases energy. (B) Explain how different factors can affect the rate of a chemical reaction.
Assignments, Labs, Homework and Study Guide to Download:
Available in word doc and in PDF
Balancing Chemical Equation Game Link
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
What happens during a chemical reaction?
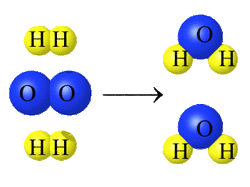
1. Bonds are broken: chemical bonds break between the atoms.
2. Atoms are rearranged: atoms will rearrange themselves.
3. New bonds are formed: new bonds form with the rearranged atoms to make
new substances.
2. Atoms are rearranged: atoms will rearrange themselves.
3. New bonds are formed: new bonds form with the rearranged atoms to make
new substances.
The 5 types of chemical reactions
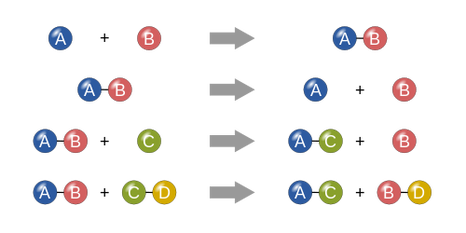
1. Synthesis : two combine to form one
2. Decomposition: One breaks down to form 2 substances
3. Single replacement: One replaces the two that are combined
4. Double replacement: the doubles switch with each other.
5. Combustion: involves oxygen and releases CO2 and H2O
Ionic Bonds - Metal and Non-metal
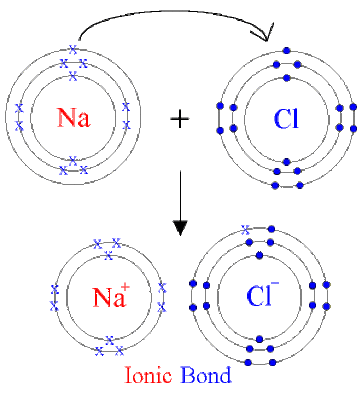
Ionic bonds happen between a Metal and a Non-metal. In this case, sodium (Na) is the metal and chlorine (Cl) is the non-metal.
Na is not happy because it wants it's outer energy level to be full, in this case, it needs 8 electrons in it. The "X" represents an electron. It first level has 2, its middle has 8 and it's third level has the one electron. It could gain 7 electrons or lose it's one electron to have a full 3rd level or a full 2nd level.
Cl is not happy because it needs one more electron to be full. It could gain one electron or lose 7 to be full.
So Na wants to lose one electron and Cl wants to gain one electron.....so they do....Na gives Cl its electron.
Na gains a charge of 1+ because it lost one negative electron (becomes a cation ion), and Cl becomes 1- because it gained an electron (becomes an anion).
In nature, opposites attract, so 1+ and 1- attract each other and form a chemical bond....this bond is a very strong ionic bond because it bonded between two ions.
Na is not happy because it wants it's outer energy level to be full, in this case, it needs 8 electrons in it. The "X" represents an electron. It first level has 2, its middle has 8 and it's third level has the one electron. It could gain 7 electrons or lose it's one electron to have a full 3rd level or a full 2nd level.
Cl is not happy because it needs one more electron to be full. It could gain one electron or lose 7 to be full.
So Na wants to lose one electron and Cl wants to gain one electron.....so they do....Na gives Cl its electron.
Na gains a charge of 1+ because it lost one negative electron (becomes a cation ion), and Cl becomes 1- because it gained an electron (becomes an anion).
In nature, opposites attract, so 1+ and 1- attract each other and form a chemical bond....this bond is a very strong ionic bond because it bonded between two ions.
Covalent Bonds - Nonmetal and nonmetal
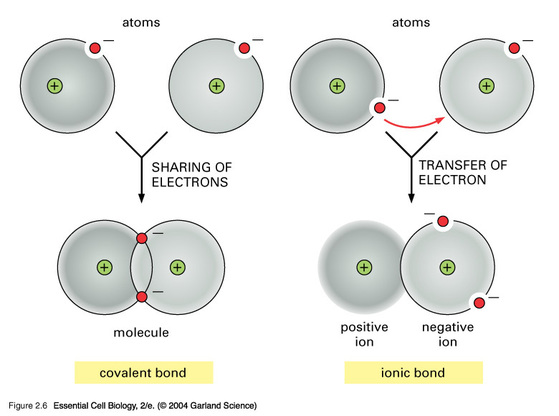
In a covalent bonds, atoms will SHARE electrons to get a full outer energy level.
In this case, two hydrogen atoms each have one electron. Hydrogen wants two electrons in it's outer energy level, which for energy level one it is two electrons.
Instead of one of them giving up their electron, the two atoms will get close enough to share their electrons, and in each case they will have two electrons in their outer energy level, making them both happy.
This sharing creates a bond, called a covalent bond.
It they share the electrons equally, it creates a non-polar bond.
If they share them unequally, it creates a polar covalent bond, usually represented by a bend in the chemical structure, but not always.
In this case, two hydrogen atoms each have one electron. Hydrogen wants two electrons in it's outer energy level, which for energy level one it is two electrons.
Instead of one of them giving up their electron, the two atoms will get close enough to share their electrons, and in each case they will have two electrons in their outer energy level, making them both happy.
This sharing creates a bond, called a covalent bond.
It they share the electrons equally, it creates a non-polar bond.
If they share them unequally, it creates a polar covalent bond, usually represented by a bend in the chemical structure, but not always.
Metallic Bonds- Metal and Metal
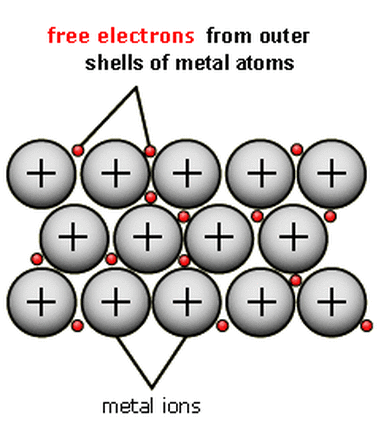
In a metallic bond, this forms between a metal and another metal. It's found in ALL metals...thus the name metallic bond.
What happens is all the metal atoms give up some of their electrons becoming cations. The electrons are then able to flow around the outer shell and through the metal. This is what gives metals their property of electric conductivity. Their ability to have electricity flow through them.
What happens is all the metal atoms give up some of their electrons becoming cations. The electrons are then able to flow around the outer shell and through the metal. This is what gives metals their property of electric conductivity. Their ability to have electricity flow through them.
Self Check Quiz:
Questions: Answers:
1. Covalent bonds are formed between (metals, non-metals or metalloids) 1. Non-metals
2. An ionic bond is a bond that forms between… 2. Non-metal & Metal
3. The forces that hold atoms and ions together are known as what? 3. Chemical bonds
4. T/F Bubbles forming is a clear sign of a physical change. 4. False
5. The kinetic theory states that molecules move faster with increased ________________. 5. energy
6. When two chlorines bond they will ____________________ valence electrons to be more stable. 6. share
7. What happens in a chemical reaction? 7. Atoms are rearranged.
8. A chemical equation is balanced by changing or adding what? 8. Coefficients
9. T/F A precipitate is a solid formed from two liquids. 9. True
10. T/F There are always the same number of atoms in reactants and products. 10. True
11. A change of ____________________ is a sign that a chemical reaction is taking place 11. color
12. When elements combine they may form a _____________________ with very different properties from the atoms making it up. 12. Compound
13. The chemical formula H2O means water has ______ hydrogen atom(s) and ______ Oxygen atom(s). 13. 2, 1
14. What happens during a chemical reaction (list the 3 steps – 3 points)? 14. Bonds are broken, atoms rearranged, new bonds formed
15. An enzyme is a special kind of catalyst that works to… 15. speed up a reaction
16. List 5 things that will speed up a reaction 16. Increase surface area, pressure, energy, concentration, & add a catalyst
17. Match the type of reaction (Single, Double, Decomposition, Synthesis, Combustion)
A) H20 ---> O2 + H2 A) Decomposition
B) CH4 + O2 + Energy ------> Heat + CO2 + H20 B) Combustion
C) Fe + CuSO4 -------> Cu + FeSO4 C) Single Replacement
D) Fe + O2 -------> FeO2 D) Synthesis
E) 2AgNO3 + CaCl2 ---------> 2AgCl + Ca(NO3)2 E) Double Replacement
Questions: Answers:
1. Covalent bonds are formed between (metals, non-metals or metalloids) 1. Non-metals
2. An ionic bond is a bond that forms between… 2. Non-metal & Metal
3. The forces that hold atoms and ions together are known as what? 3. Chemical bonds
4. T/F Bubbles forming is a clear sign of a physical change. 4. False
5. The kinetic theory states that molecules move faster with increased ________________. 5. energy
6. When two chlorines bond they will ____________________ valence electrons to be more stable. 6. share
7. What happens in a chemical reaction? 7. Atoms are rearranged.
8. A chemical equation is balanced by changing or adding what? 8. Coefficients
9. T/F A precipitate is a solid formed from two liquids. 9. True
10. T/F There are always the same number of atoms in reactants and products. 10. True
11. A change of ____________________ is a sign that a chemical reaction is taking place 11. color
12. When elements combine they may form a _____________________ with very different properties from the atoms making it up. 12. Compound
13. The chemical formula H2O means water has ______ hydrogen atom(s) and ______ Oxygen atom(s). 13. 2, 1
14. What happens during a chemical reaction (list the 3 steps – 3 points)? 14. Bonds are broken, atoms rearranged, new bonds formed
15. An enzyme is a special kind of catalyst that works to… 15. speed up a reaction
16. List 5 things that will speed up a reaction 16. Increase surface area, pressure, energy, concentration, & add a catalyst
17. Match the type of reaction (Single, Double, Decomposition, Synthesis, Combustion)
A) H20 ---> O2 + H2 A) Decomposition
B) CH4 + O2 + Energy ------> Heat + CO2 + H20 B) Combustion
C) Fe + CuSO4 -------> Cu + FeSO4 C) Single Replacement
D) Fe + O2 -------> FeO2 D) Synthesis
E) 2AgNO3 + CaCl2 ---------> 2AgCl + Ca(NO3)2 E) Double Replacement