The Atom and Periodic Table
This unit is about the parts of an atom (protons, neutrons, and electrons. Along with the parts, we will learn about how atoms are arranged in the periodic table (periodic law), how this organization creates many patterns.
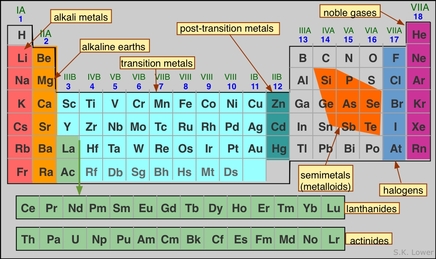
Some Patterns in the Periodic Table:
Each atom increases by one proton as you move across a row/period.
Each atom increases by one electron as you move across a row/period.
The mass of atoms increase as you move across a row/period.
Metals are located on the left/middle of the periodic table.
Nonmetals are located on the right side of the table.
Metalloids are in between the metals and nonmetals.
Each row/period starts when an electron fills a new energy level.
Each row/period ends when an energy level is full.
State Standards Covered:
H.1P.1 (A) Explain how atomic structure is related to the properties of elements and their position in the Periodic Table.
Some Patterns in the Periodic Table:
Each atom increases by one proton as you move across a row/period.
Each atom increases by one electron as you move across a row/period.
The mass of atoms increase as you move across a row/period.
Metals are located on the left/middle of the periodic table.
Nonmetals are located on the right side of the table.
Metalloids are in between the metals and nonmetals.
Each row/period starts when an electron fills a new energy level.
Each row/period ends when an energy level is full.
State Standards Covered:
H.1P.1 (A) Explain how atomic structure is related to the properties of elements and their position in the Periodic Table.
Assignments, Labs, Homework and Study Guide to Download:
Available in word doc and in PDF
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Self Check
1. What are the 3 sub atomic particles in an atom? Answer: Protons, Neutrons, and Electrons
2. An atom with group/family 1 has how many valence electrons? Answers: 1
3. What is a valence electron? Answer: Electrons in the outer energy level.
4. How do the number of protons/electrons change as you move left to right on the periodic table? Answer: Increase by one.
12 (2...8....2 listed on the side)
Mg
Magnesium
24.3
5. How many protons are in the above element? Answer: 12
6. How many electrons? Answer: 12
7. How many neutrons? Answer: 12 (24-12)
8. How many electrons in each orbit? Answer: 2 in 1st, 8 in 2nd, 2 in 3rd.
9. Family or group that it is in ? Answer: Family/group #2
10. Period or Row that it is in? Answer: Period/family #3
11. Most elements on the periodic table are _____________? Answer: Metals
12. Metals are located on the _____ side and _________ of the periodic table. Answer: left, middle
13. The periodic table is arranged by.............. Answer: Increasing # of protons.
14. A positive ion is a _________ and a negative ion is an ____________. Answer: Cation, Anion
15. Isotopes are atoms with extra ____________ . Answer: neutrons
16. Can you name the atom in the heading on this page (left side). Answer: Magnesium
17. What makes an element reactive or stable? Answer: If all the valence electrons are filled it is stable, if the outer energy level
is almost full (missing 1) or has only 1 valence electron, it makes it reactive.
18. How is the periodic table arranged? (Use protons, valence electrons and energy levels)
Answer: The ORDER of the the periodic table is determined by the number of protons in the atom.
Each row on the periodic table is an energy level, ex: row 3 all have 3 energy levels.
The families/groups on the table all have the same number of valence electrons.
2. An atom with group/family 1 has how many valence electrons? Answers: 1
3. What is a valence electron? Answer: Electrons in the outer energy level.
4. How do the number of protons/electrons change as you move left to right on the periodic table? Answer: Increase by one.
12 (2...8....2 listed on the side)
Mg
Magnesium
24.3
5. How many protons are in the above element? Answer: 12
6. How many electrons? Answer: 12
7. How many neutrons? Answer: 12 (24-12)
8. How many electrons in each orbit? Answer: 2 in 1st, 8 in 2nd, 2 in 3rd.
9. Family or group that it is in ? Answer: Family/group #2
10. Period or Row that it is in? Answer: Period/family #3
11. Most elements on the periodic table are _____________? Answer: Metals
12. Metals are located on the _____ side and _________ of the periodic table. Answer: left, middle
13. The periodic table is arranged by.............. Answer: Increasing # of protons.
14. A positive ion is a _________ and a negative ion is an ____________. Answer: Cation, Anion
15. Isotopes are atoms with extra ____________ . Answer: neutrons
16. Can you name the atom in the heading on this page (left side). Answer: Magnesium
17. What makes an element reactive or stable? Answer: If all the valence electrons are filled it is stable, if the outer energy level
is almost full (missing 1) or has only 1 valence electron, it makes it reactive.
18. How is the periodic table arranged? (Use protons, valence electrons and energy levels)
Answer: The ORDER of the the periodic table is determined by the number of protons in the atom.
Each row on the periodic table is an energy level, ex: row 3 all have 3 energy levels.
The families/groups on the table all have the same number of valence electrons.